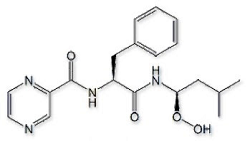
Bortezomib USP Impurity L
1 Results Found
Page 1 of 1
Bortezomib API Reference standards and its EP Impurities, USP Impurities, non pharmacopeial Impurities, listed below & also offered custom synthesis for unknown Impurities. If require any contact info@drjcrbio.com, sales@drjcrbio.com
-
Free
Product Name : Bortezomib USP Impurity L ; Bortezomib R-Hydroxyperoxide (USP) ; (R)-Hydroperoxy Des(boric Acid) Bortezomib ; N-((S)-1-(((R)-1-Hydroperoxy-3-methylbutyl)amino)-1-oxo-3-phenylpropan-2-yl)pyrazine-2-carboxamide | 886979-78-8
Code: JCBRZ-18 CAS: 886979-78-8 Chemical Formula: C19H24N4O4 Molecular Weight: 372.42 Category: Reference Standards/API Drug Metabolites/EP Impurities/USP Impurities Literature: Application: Bortezomib USP Impurity L ; Bortezomib R-Hydroxyperoxide (USP) ; (R)-Hydroperoxy Des(boric Acid) Bortezomib ; N-((S)-1-(((R)-1-Hydroperoxy-3-methylbutyl)amino)-1-oxo-3-phenylpropan-2-yl)pyrazine-2-carboxamide | 886979-78-8is useful as reference Impurity standard.